|
5/9/2023 0 Comments Lumo and humo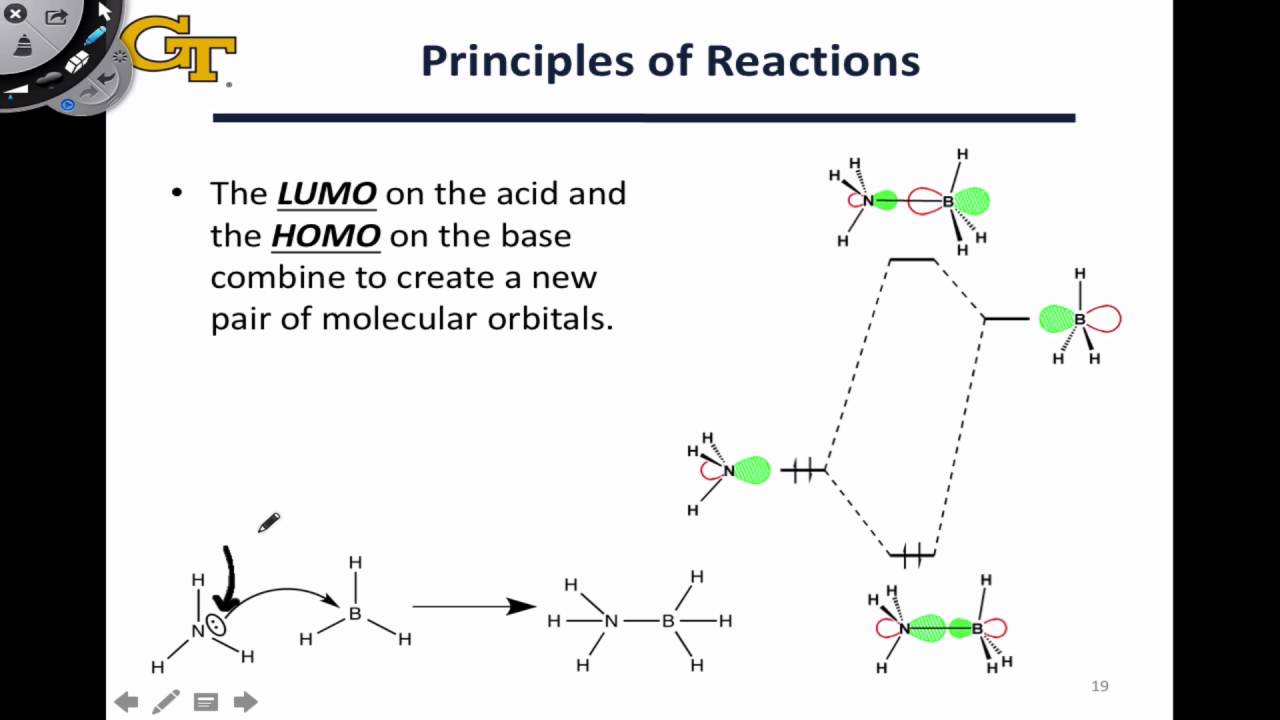
Thus, red represents negative regions and blue represents positive regions on an electrostatic potential surface. The electrostatic potential is typically "painted" on the electron density isosurface.īy default WebMO represents smaller values by red and larger values in blue. The electrostatic potential is the energy that a unit charge at each point in space would have due to the surrounding electrons and nuclei, which is computed by integrating the electron density divided by distance at each point in space. The electron density isosurface represents the size and shape of the molecule. The electron density is computed from the sum of squares of occupied molecular orbitals. Interpreting Molecular Orbital Imagesīy default WebMO represents occupied orbitals in red/blue and unoccupied orbitals in yellow/green. When the Orbitals list is displayed in the Molecule Viewer, clicking on any particular orbital will display it. The orbital zoom is controlled by choosing View:Zoome ( ) and dragging vertically. The location of the orbital is controlled by choosing View:Translate ( ) and dragging vertically. The orientation of the orbital is controlled by choosing View:Rotate ( ) and dragging in the x-y plane or alt-dragging horizontally for z-axis rotation. The orbital definitions (basis functions and linear combination coefficients) may be saved locally by clicking the Download icon ( ). The electron density, electrostatic potential, and electrophilic/nucleophilic/radical frontier densities may also be displayed. Individual orbitals may be displayed by clicking the corresponding View icon ( ). The symmetry, occupancy, and energy of each orbital is tablulated in the Molecular Orbitals section of Calculated Quantities. Molecular orbitals and related quantities are displayed on the View Results page in the Molecule Viewer tab of the 3-D visualizer. Hence, smaller HOMO-LUMO gaps correspond to better stability.When a job has completed, from the Job Manager page click either the job name or the View Job icon ( ). The greater the mobility of the #pi# electrons in large conjugated pi orbital systems, the greater the distribution of the energy throughout the molecule, stabilizing it. When you have a large aromatic system in particular, small HOMO-LUMO gaps lead to mobile #\mathbf(pi)# electrons since it is easy for the electron to jump to a higher energy level that is close in energy. 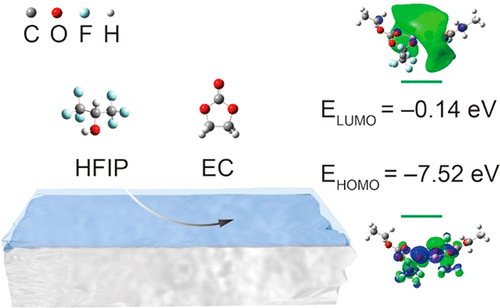
The larger the aromatic system is, the smaller the HOMO-LUMO gap! Hence, it is the most important energy gap to consider.Įxcitations becomes easier as the HOMO-LUMO gap converges, such as for large aromatic systems (like tetracene or benzopyrene), or for transition-metal complexes (that is why they tend to be colored). That means they are closest in energy out of all orbitals in the molecule.ĭue to the energies of these orbitals being the closest of any orbitals of different energy levels, the HOMO-LUMO gap is where the most likely excitations can occur. 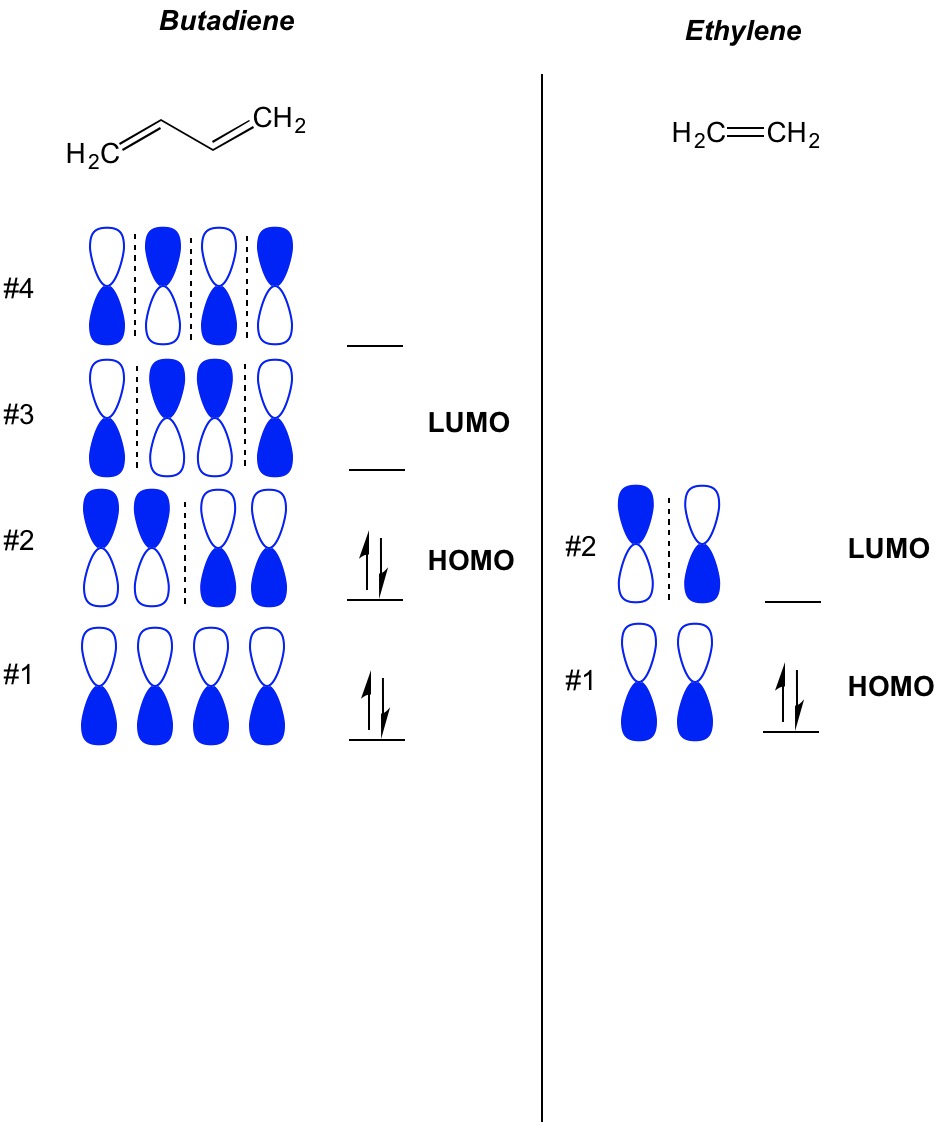
Of the orbitals that have electrons, the HOMO is the highest in energy, and of the orbitals that don't, the LUMO is the lowest in energy. Don't be confused though, because the LUMO is higher in energy than the HOMO. HOMO stands for " Highest Occupied Molecular Orbital", and LUMO stands for " Lowest Unoccupied Molecular Orbital".
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
